 After cannulation and securement, the inserter (E.D.) confirmed functionality with blood sampling (10 mL) and flushing without resistance. The inserter captured digital images with measurements of vessel depth and diameter. 
The proceduralist (E.D.) performed the insertion with ultrasound guidance. 3, 15, 16 Given the high risk in 1 protected catheter, we aimed to evaluate whether the thrombosis risk can be mitigated by using an alternative MC with a distinct antithrombotic mechanism: a chlorhexidine gluconate coating that reduces fibrin sheath development in in vivo ovine models. 3, 14, 15, 16 Despite this enhancement, these MCs demonstrated a CRT risk that was substantially higher than even that of central venous access devices (CVADs). 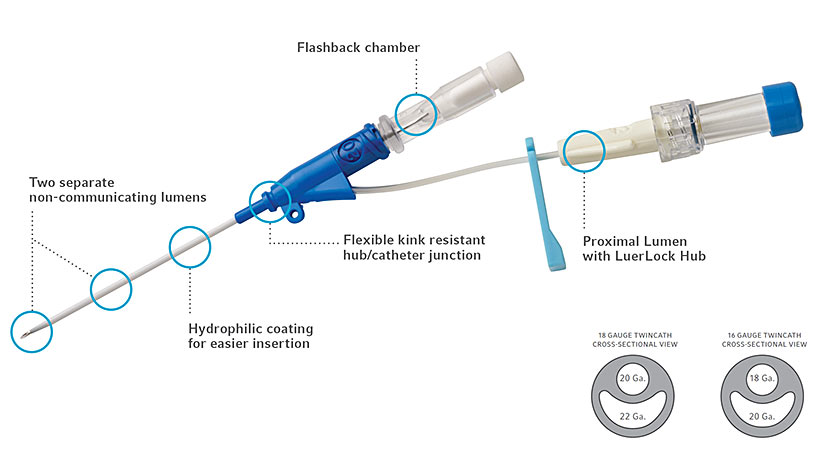
All catheters in this cohort were manufactured with the addition of a polymer to the base polyurethane that was found to reduce thrombosis in vitro. In 2019, a large retrospective analysis 13 of 1094 MCs found that 12% were associated with symptomatic CRT. 9, 10, 11 The current data on this complication remain extremely limited, equivocal, and highly variable, with rates of symptomatic CRT ranging from 0% to 12%. Thrombosis interrupts and delays venous therapy, increases cost of care, and can lead to significant adverse patient outcomes, such as bloodstream infection, pulmonary embolism, and postthrombotic syndrome. Symptomatic CRT is one of the most serious complications of catheter insertion. 2, 4, 5, 6 Despite the rapid expansion of MCs across the globe, few randomized clinical trials and limited high-quality prospective investigations exist that explore complication risks, including catheter-associated bloodstream infection (CABSI) and catheter-related thromboembolism (CRT). 2, 3 These recommendations are largely based on expert opinion focussing on the functionality of MCs rather than the risks of these devices. 1 Guidelines highlight MCs as the ideal choice for intermediate duration of therapy and the best choice for many patients with difficult vascular access (DVA). Midline catheters (MCs) have rapidly infiltrated the marketplace since 2015, with nearly 850 000 units sold in the US in 2019. Role of the Funder/Sponsor: The funding source had no role in the design and conduct of the study collection, management, analysis, and interpretation of the data preparation, review, or approval of the manuscript and the decision to submit the manuscript for publication.ĭata Sharing Statement: See Supplement 3. No other disclosures were reported.įunding/Support: The research was funded via an educational grant for research from Teleflex. Ms Diloreto reported receiving grants from Teleflex and BD during the conduct of the study outside the submitted work. Corresponding Author: Amit Bahl, MD, MPH, Department of Emergency Medicine, Beaumont Hospital, Royal Oak, 3601 13 Mile Rd, Royal Oak, MI 48073 ( Contributions: Dr Bahl had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.Īcquisition, analysis, or interpretation of data: All authors.ĭrafting of the manuscript: Bahl, Diloreto, Hijazi, Chen.Ĭritical revision of the manuscript for important intellectual content: Bahl, Diloreto, Jankowski, Chen.Īdministrative, technical, or material support: Bahl, Diloreto, Jankowski.Ĭonflict of Interest Disclosures: Dr Bahl reported receiving grants from Teleflex during the conduct of the study and personal fees from the Teleflex Key Opinion Leader program outside the submitted work. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
